April 1, 2020
MUSTANG: The drug discovery platform
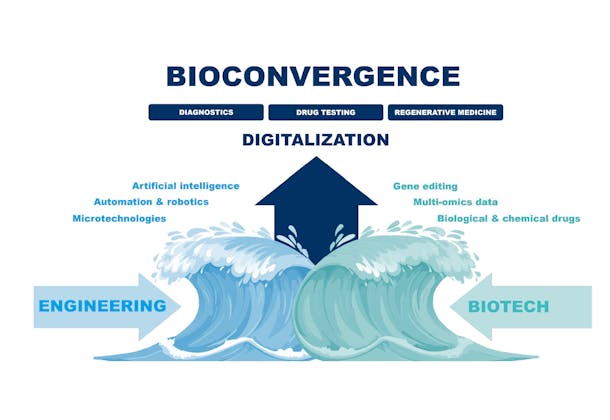
The novel screening platform ‘MUSTANG’ (Muscle Tissue Analyzer Gear) is a first-of-its-kind automated solution capable of measuring the contractility (strength) of 3D printed micro-muscles via electrical pulse stimulation. Developed by Novartis, CSEM, Life Imaging Services, Weidmann Medical Technology and the Zurich University of Applied Sciences (ZHAW), the apparatus is set to enhance the reliability and robustness of drug development for Musculoskeletal Diseases.